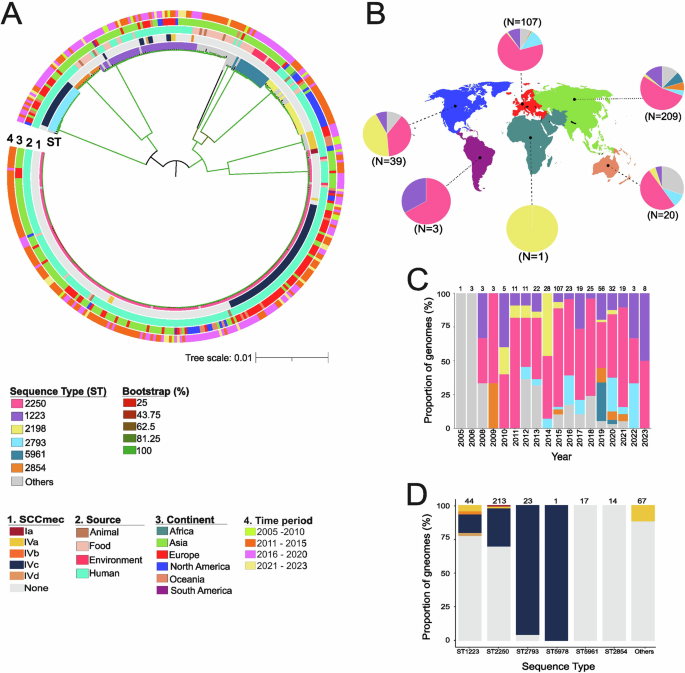
STgen Bio announced Thursday that it will expand its first plant to meet rising demand for high-titer biopharmaceuticals.
This capacity boost involves adding midsize facilities suitable for multiproduct production, aimed at expanding the company’s business portfolio and securing global orders.
The investment of approximately 110 billion won ($77 million) will be used to expand drug substance and drug product production facilities and establish related infrastructure. Production capacity will increase from 9,000 liters to 14,000 liters upon the project’s completion in the first quarter of 2028.
The company will install two bioreactors and one harvester, enhancing production efficiency. Notably, the deployment of an isolator-type drug product filling line will ensure a fully aseptic environment by eliminating manual intervention. This is a strategic investment aimed at meeting the rising standards of global clients.
STgen Bio has obtained regulatory approvals from 10 countries, including the US Food and Drug Administration and the European Medicines Agency.
“Based on our compliance with global regulatory standards and our experience in commercialization, we plan to focus on high-value-added products,” an STgen Bio official said. “By advancing our production capabilities, we will secure both stability and scalability, and flexibly respond to our global clients.”
stlee0329@heraldcorp.com