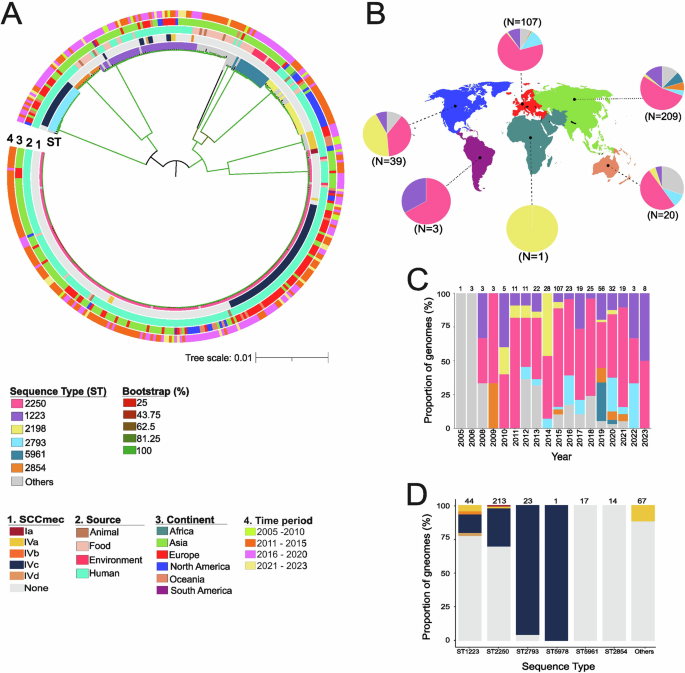
Tong, S. Y. C. et al. Virulence of endemic nonpigmented northern Australian Staphylococcus aureus clone (clonal complex 75, S. argenteus) is not augmented by staphyloxanthin. J. Infect. Dis. 208, 520–527 (2013).
Tong, S. Y. C. et al. Novel staphylococcal species that form part of a Staphylococcus aureus-related complex: the non-pigmented Staphylococcus argenteus sp. nov. and the non-human primate-associated Staphylococcus schweitzeri sp. nov. Int J. Syst. Evol. Microbiol. 65, 15–22 (2015).
Wakabayashi, Y. et al. Staphylococcal food poisoning caused by Staphylococcus argenteus harboring staphylococcal enterotoxin genes. Int J. Food Microbiol. 265, 23–29 (2018).
Monecke, S. et al. Characterisation of PVL-Positive Staphylococcus argenteus from the United Arab Emirates. Antibiotics 13, 401 (2024).
Supriadi, I. R. et al. Identification and characterization of Staphylococcus argenteus from Indonesia. Int. J. Med. Microbiol. 316, 151629 (2024).
Chen, S.-Y. et al. High mortality impact of Staphylococcus argenteus on patients with community-onset staphylococcal bacteraemia. Int J. Antimicrob. Agents 52, 747–753 (2018).
Lee, M. et al. Staphylococcus argenteus bacteremia in the Republic of Korea. Microbiol. Spectr. 12, e0279823 (2024).
Rigaill, J. et al. Community-acquired Staphylococcus argenteus sequence type 2250 bone and joint infection, France, 2017. Emerg. Infect. Dis. 24, 1958–1961 (2018).
Ohnishi, T. et al. Purulent lymphadenitis caused by Staphylococcus argenteus, representing the first Japanese case of Staphylococcus argenteus (multilocus sequence type 2250) infection in a 12-year-old boy. J. Infect. Chemother. 24, 925–927 (2018).
Rong, D. et al. Prevalence and characterization of Staphylococcus aureus and Staphylococcus argenteus isolated from rice and flour products in Guangdong, China. Int. J. Food Microbiol. 406, 110348 (2023).
Indrawattana, N. et al. Staphylococcus argenteus from rabbits in Thailand. Microbiologyopen 8, e00665 (2019).
Meijer, E. F. J. et al. Canine Staphylococcus argenteus: case report from The Netherlands. Pathogens 11, 153 (2022).
Hao, S. et al. Genomic profiling of evolving daptomycin resistance in a patient with recurrent Staphylococcus argenteus sepsis. Antimicrob. Agents Chemother. 64, e00961-20 (2020).
Shittu, A. O. et al. First report of a methicillin-resistant, high-level mupirocin-resistant Staphylococcus argenteus. Front. Cell Infect. Microbiol. 12, 860163 (2022).
Witteveen, S. et al. Genetic characteristics of methicillin-resistant Staphylococcus argenteus isolates collected in the Dutch National MRSA Surveillance from 2008 to 2021. Microbiol. Spectr. 10, e0103522 (2022).
Giske, C. G. et al. Transmission events and antimicrobial susceptibilities of methicillin-resistant Staphylococcus argenteus in Stockholm. Clin. Microbiol. Infect. 25, 1289.e5–1289.e8 (2019).
Jauneikaite, E. et al. Staphylococcus argenteus transmission among healthy Royal Marines: a molecular epidemiology case-study. J. Infect. 83, 550–553 (2021).
Wu, S. et al. Evolutionary divergence of the novel Staphylococcal species Staphylococcus argenteus. Front. Microbiol. 12, 769642 (2021).
Eshaghi, A. et al. Phenotypic and genomic profiling of Staphylococcus argenteus in Canada and the United States and recommendations for clinical result reporting. J. Clin. Microbiol. 59, e02470-20 (2021).
Goswami, C., Fox, S., Holden, M., Leanord, A. & Evans, T. J. Genomic analysis of global Staphylococcus argenteus strains reveals distinct lineages with differing virulence and antibiotic resistance gene content. Front. Microbiol. 12, 795173 (2021).
Moradigaravand, D. et al. Evolution of the Staphylococcus argenteus ST2250 clone in northeastern Thailand is linked with the acquisition of livestock-associated Staphylococcal genes. mBio 8, e00802–17 (2017).
Yan, A., Kus, J. V. & Sant, N. The Staphylococcus aureus complex: implications for the clinical microbiology laboratory. J. Clin. Microbiol. 63, e0127624 (2025).
Kislyuk, A. O., Haegeman, B., Bergman, N. H. & Weitz, J. S. Genomic fluidity: an integrative view of gene diversity within microbial populations. BMC Genom. 12, 32 (2011).
Tettelin, H., Riley, D., Cattuto, C. & Medini, D. Comparative genomics: the bacterial pan-genome. Curr. Opin. Microbiol. 11, 472–477 (2008).
Liu, J. et al. Staphylococcal chromosomal cassettes mec (SCCmec): a mobile genetic element in methicillin-resistant Staphylococcus aureus. Micro. Pathog. 101, 56–67 (2016).
International Working Group on the Classification of Staphylococcal Cassette Chromosome Elements (IWG-SCC) Classification of staphylococcal cassette chromosome mec (SCCmec): guidelines for reporting novel SCCmec elements. Antimicrob. Agents Chemother. 53, 4961–4967 (2009).
Uehara, Y. Current status of Staphylococcal Cassette Chromosome mec (SCCmec). Antibiotics 11, 86 (2022).
Horvath, E. R. B., Stein, M. G., Mulvey, M. A., Hernandez, E. J. & Winter, J. M. Resistance Gene Association and Inference Network (ReGAIN): a bioinformatics pipeline for assessing probabilistic co-occurrence between resistance genes in bacterial pathogens. Preprint at https://doi.org/10.1101/2024.02.26.582197 (2024).
Suzuki, Y. et al. Food poisoning outbreak in Tokyo, Japan caused by Staphylococcus argenteus. Int. J. Food Microbiol. 262, 31–37 (2017).
Cavaiuolo, M. et al. First report of enterotoxigenic Staphylococcus argenteus as a foodborne pathogen. Int. J. Food Microbiol. 394, 110182 (2023).
Chambers, H. F. & Deleo, F. R. Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat. Rev. Microbiol. 7, 629–641 (2009).
Hendlin, D. et al. Phosphonomycin, a new antibiotic produced by strains of Streptomyces. Science 166, 122–123 (1969).
Matthews, P. C. et al. Oral fosfomycin for treatment of urinary tract infection: a retrospective cohort study. BMC Infect. Dis. 16, 556 (2016).
Omori, K. et al. Fosfomycin as salvage therapy for persistent methicillin-resistant Staphylococcus aureus bacteremia: a case series and review of the literature. J. Infect. Chemother. 30, 352–356 (2024).
Turner, N. A. et al. Methicillin-resistant Staphylococcus aureus: an overview of basic and clinical research. Nat. Rev. Microbiol. 17, 203–218 (2019).
Monte, D. F. M. & de Oliveira, C. J. B. Global trends in the increasing prevalence of the fosfomycin resistance gene in Staphylococcus aureus. Lancet Microbe 5, e104 (2024).
Etienne, J., Gerbaud, G., Courvalin, P. & Fleurette, J. Plasmid-mediated resistance to fosfomycin in Staphylococcus epidermidis. FEMS Microbiol. Lett. 52, 133–137 (1989).
McClure, J.-A. et al. Staphylococcus aureus ST59: concurrent but separate evolution of North American and East Asian lineages. Front. Microbiol. 12, 631845 (2021).
Hall, M. D., Woolhouse, M. E. J. & Rambaut, A. The effects of sampling strategy on the quality of reconstruction of viral population dynamics using Bayesian skyline family coalescent methods: a simulation study. Virus Evol. 2, vew003 (2016).
Arias, C. A. et al. A prospective cohort multicenter study of molecular epidemiology and phylogenomics of Staphylococcus aureus bacteremia in nine Latin American countries. Antimicrob. Agents Chemother. 61, e00816-17 (2017).
Hisatsune, J. et al. Staphylococcus aureus ST764-SCCmecII high-risk clone in bloodstream infections revealed through national genomic surveillance integrating clinical data. Nat. Commun. 16, 2698 (2025).
Choi, S. H. et al. A longitudinal study of adult patients with Staphylococcus aureus bacteremia over 11 years in Korea. J. Korean Med. Sci. 36, e104 (2021).
Chen, H. et al. Drivers of methicillin-resistant Staphylococcus aureus (MRSA) lineage replacement in China. Genome Med. 13, 171 (2021).
Souza, S. S. R. et al. Demographic fluctuations in bloodstream Staphylococcus aureus lineages configure the mobile gene pool and antimicrobial resistance. NPJ Antimicrob. Resist. 2, 14 (2024).
Méric, G. et al. Ecological overlap and horizontal gene transfer in Staphylococcus aureus and Staphylococcus epidermidis. Genome Biol. Evol. 7, 1313–1328 (2015).
Kastman, E. K. et al. Biotic interactions shape the ecological distributions of Staphylococcus species. mBio 7, e01157-16 (2016).
Ikhimiukor, O. O. et al. Leaky barriers to gene sharing between locally co-existing coagulase-negative Staphylococcus species. Commun. Biol. 6, 482 (2023).
Souvorov, A., Agarwala, R. & Lipman, D. J. SKESA: strategic k-mer extension for scrupulous assemblies. Genome Biol. 19, 153 (2018).
Gurevich, A., Saveliev, V., Vyahhi, N. & Tesler, G. QUAST: quality assessment tool for genome assemblies. Bioinformatics 29, 1072–1075 (2013).
Parks, D. H., Imelfort, M., Skennerton, C. T., Hugenholtz, P. & Tyson, G. W. CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 25, 1043–1055 (2015).
Jain, C., Rodriguez-R, L. M., Phillippy, A. M., Konstantinidis, K. T. & Aluru, S. High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries. Nat. Commun. 9, 5114 (2018).
Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics 30, 2068–2069 (2014).
Tonkin-Hill, G. et al. Producing polished prokaryotic pangenomes with the Panaroo pipeline. Genome Biol. 21, 180 (2020).
Medini, D., Donati, C., Tettelin, H., Masignani, V. & Rappuoli, R. The microbial pan-genome. Curr. Opin. Genet Dev. 15, 589–594 (2005).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Page, A. J. et al. SNP-sites: rapid efficient extraction of SNPs from multi-FASTA alignments. Micro. Genom. 2, e000056 (2016).
Minh, B. Q. et al. IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 37, 1530–1534 (2020).
Tavaré, S. Some probabilistic and statistical problems in the analysis of DNA sequences. Lect. Math. Life Sci. 17, 57–86 (1986).
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K. F., von Haeseler, A. & Jermiin, L. S. ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Methods 14, 587–589 (2017).
Letunic, I. & Bork, P. Interactive Tree of Life (iTOL) v6: recent updates to the phylogenetic tree display and annotation tool. Nucleic Acids Res. 52, W78–W82 (2024).
Jolley, K. A., Bray, J. E. & Maiden, M. C. J. Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res. 3, 124 (2018).
Enright, M. C., Day, N. P., Davies, C. E., Peacock, S. J. & Spratt, B. G. Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J. Clin. Microbiol. 38, 1008–1015 (2000).
Alcock, B. P. et al. CARD 2023: expanded curation, support for machine learning, and resistome prediction at the comprehensive antibiotic resistance database. Nucleic Acids Res. 51, D690–D699 (2023).
Liu, B., Zheng, D., Zhou, S., Chen, L. & Yang, J. VFDB 2022: a general classification scheme for bacterial virulence factors. Nucleic Acids Res. 50, D912–D917 (2022).
Petit, R. A. & Read, T. D. Staphylococcus aureus viewed from the perspective of 40,000+ genomes. PeerJ 6, e5261 (2018).
Carattoli, A. et al. In silico detection and typing of plasmids using PlasmidFinder and plasmid multilocus sequence typing. Antimicrob. Agents Chemother. 58, 3895–3903 (2014).
RStudio Team. RStudio: Integrated Development for R. http://www.rstudio.com/ (2020).
Ondov, B. D. et al. Mash: fast genome and metagenome distance estimation using MinHash. Genome Biol. 17, 132 (2016).
Wickham, H. et al. Welcome to the Tidyverse. J. Open Source Softw. 4, 1686 (2019).
Croucher, N. J. et al. Rapid phylogenetic analysis of large samples of recombinant bacterial whole genome sequences using Gubbins. Nucleic Acids Res. 43, e15 (2015).
Schwarz, G. Estimating the dimension of a model. Ann. Stat. 6, 461–464 (1978).
Rambaut, A., Lam, T. T., Max Carvalho, L. & Pybus, O. G. Exploring the temporal structure of heterochronous sequences using TempEst (formerly Path-O-Gen). Virus Evol. 2, vew007 (2016).
Didelot, X., Croucher, N. J., Bentley, S. D., Harris, S. R. & Wilson, D. J. Bayesian inference of ancestral dates on bacterial phylogenetic trees. Nucleic Acids Res. 46, e134 (2018).
Wickham, H., François, R., Henry, L., Müller, K. & Vaughan, D. dplyr: a grammar of data manipulation. R package version 1.1.4 https://dplyr.tidyverse.org (2025).
Grolemund, G. & Wickham, H. Dates and times made easy with lubridate. J. Stat. Softw. 40, 1–25 (2011).
R Core Team. R: A language and environment for statistical computing https://www.R-project.org/ (2025).
Plummer, M., Best, N., Cowles, K. & Vines, K. CODA: convergence diagnosis and output analysis for MCMC. R. N. 6, 7–11 (2006).
Spiegelhalter, D. J., Best, N. G., Carlin, B. P. & Van Der Linde, A. Bayesian measures of model complexity and fit. J. R. Stat. Soc. Ser. B Stat. Methodol. 64, 583–639 (2002).
Gelman, A. & Rubin, D. B. Inference from iterative simulation using multiple sequences. Stat. Sci. 7, 457–472 (1992).
Brooks, S. P. & Gelman, A. General methods for monitoring convergence of iterative simulations. J. Comput. Graph. Stat. 7, 434–455 (1998).
Volz, E. M. & Didelot, X. Modeling the growth and decline of pathogen effective population size provides insight into epidemic dynamics and drivers of antimicrobial resistance. Syst. Biol. 67, 719–728 (2018).
Patil, I. Visualizations with statistical details: the ‘ggstatsplot’ approach. J. Open Source Softw. 6, 3167 (2021).